Very high active ingredient content and crosslinking for less pain
Compared to other single-injection products, MONOVISC® is supported by a large placebo-controlled, randomized, double-blind, multicenter study. This FDA approval study shows: MONOVISC® achieves the greatest reduction in pain compared to baseline, with a long-lasting effect. MONOVISC® relieves pain in knee osteoarthritis for a very long period of at least six months.
Effective
- Single-injection therapy with advanced crosslinking technology for long-lasting residence time in the joint
- Very high hyaluronic acid active ingredient content in just one injection (88 mg)
- Proven efficacy and safety confirmed by an FDA approval study
Safe
- Biotechnological manufacturing process through fermentation; free from animal proteins
- Extremely high level of purity
- Production of the highly purified and sterile hyaluronic acid solution in Boston, USA
Patient-friendly
- Only 1 injection per treatment cycle
- Fast and long-lasting pain relief as well as improved joint mobility
- Less injection-related stress/pain for the patient
Optimally tolerated, manufactured using a patented biotechnological process
- The six-month FDA approval study involving 369 patients confirms: MONOVISC® has a low rate of side effects. Only 2.2% mild reactions at the injection site were reported.5 In comparison, another single-injection product with a higher molecular weight shows a side-effect rate of 5.7%.6
- MONOVISC® has an exceptionally high level of purity and is biotechnologically produced through fermentation.
- MONOVISC® contains a highly pure and sterile hyaluronic acid solution. Thanks to its sterile packaging, MONOVISC® is suitable for use in the operating room.
- MONOVISC® is produced in Boston, USA.
PROVEN EFFECTIVENESS – Fast and long-lasting pain relief
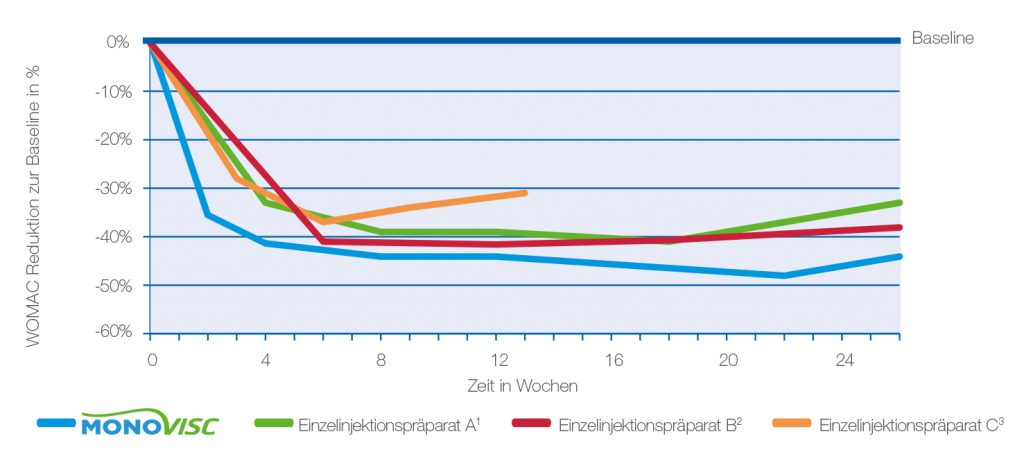
Fig. 1: Reduction of the WOMAC pain score in % over the respective study duration
- MONOVISC® reduces the WOMAC pain score by an average of 45% within six months compared to baseline.
- MONOVISC® demonstrates superior effectiveness in relieving pain in knee osteoarthritis compared to single-injection products with lower active ingredient content. 1,2,3
- MONOVISC® provides faster pain relief compared to other single-injection products: the WOMAC pain score decreases by 36% after just two weeks.
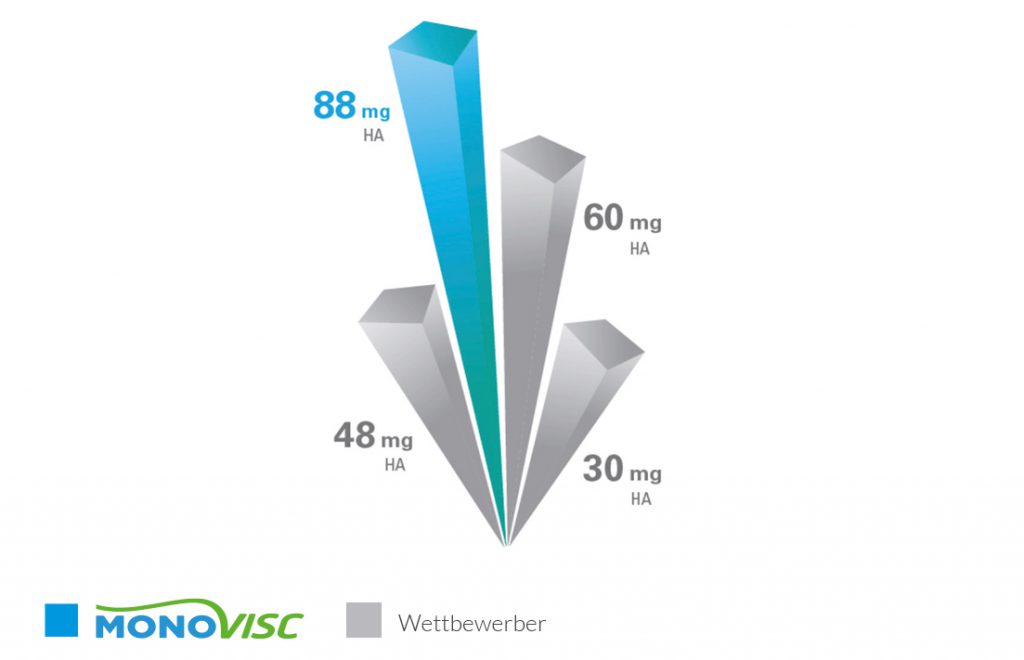
Fig. 2: Overview of crosslinked hyaluronic acid single-injection products and their hyaluronic acid content
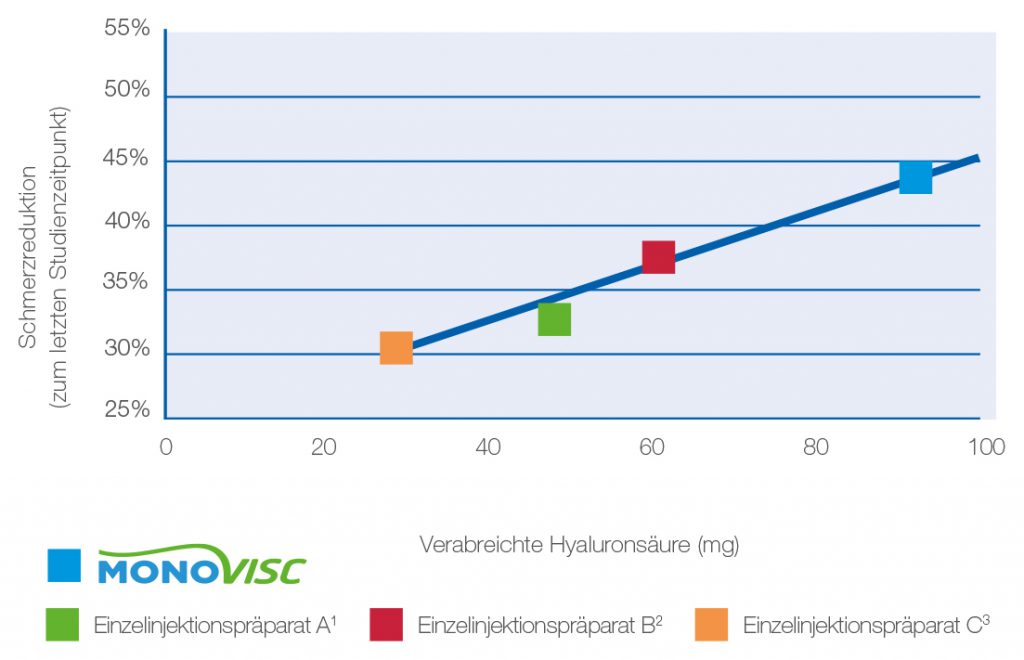
Fig. 3: Reduction of the WOMAC pain score depending on the amount of hyaluronic acid used.5
MONOVISC® delivers a high active ingredient content compared to other crosslinked one-shot products available on the market. One injection of 4 ml contains 88 mg of hyaluronic acid at a concentration of 22 mg/ml.
Minimized treatment cycle
- Since MONOVISC® requires only a single injection per treatment cycle, this means less injection-related stress/pain for the patient.
- MONOVISC® is suitable for all patients who have little time for repeated injections and who value fast and long-lasting effectiveness in their treatment.
- MONOVISC® features an ergonomically optimized syringe for safe and easy injection.
Hyaluronic acid content: 88 mg
Treatment: 1 injection
Content: 1 prefilled syringe (4 ml)
Manufacturing process: Fermentation
Production: Boston, USA
Applications: Approved for the knee, hip, shoulder, and ankle joints
Approval: Medical device with CE marking, MDR approval, and FDA approval

More Knowledge, More Movement
Injecting exogenous high–molecular weight hyaluronic acid into the joint can interrupt the osteoarthritic cascade by 1. preventing the production of inflammatory cytokines and enzymes, 2. restoring the production of natural hyaluronic acid, and 3. slowing the progression of osteoarthritis. These effects depend on the molecular weight. There is an optimal molecular weight range that maximizes hyaluronic acid’s ability to interact with the cell surface receptors of synovial cells. This interaction increases the synthesis of natural hyaluronic acid.
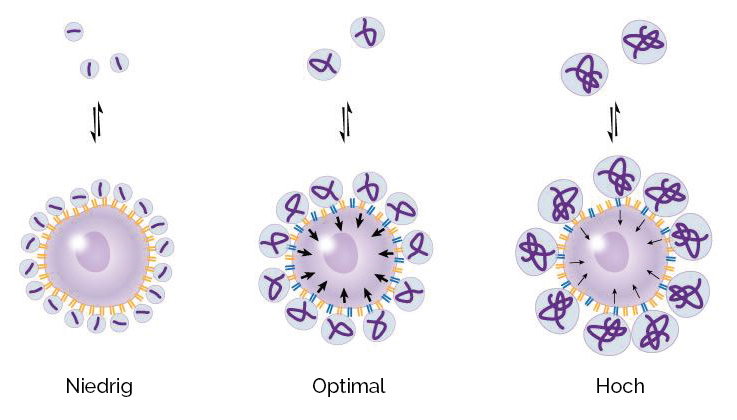
Fig. 4: Effect of hyaluronic acid molecular weight on the interaction with cell surface receptors in osteoarthritic synovial cells
- Hyaluronic acid molecules with low molecular weight bind only weakly to surface receptors. This results in little to no stimulation of hyaluronic acid biosynthesis.
- Hyaluronic acid molecules with excessively high molecular weight (> 4 million Daltons) cannot adequately bind to surface receptors due to their size. As a result, their ability to stimulate hyaluronic acid biosynthesis is limited.
- Hyaluronic acid molecules with an optimal molecular weight (between 500 thousand and 4 million Daltons) bind excellently to surface receptors, thereby maximizing the stimulation of natural hyaluronic acid biosynthesis.
MONOVISC® is manufactured using a patented crosslinking process and features a lightly crosslinked molecular structure. The resulting stabilized hyaluronic acid is broken down more slowly and remains in the joint for a longer period of time. MONOVISC® has a primary effect as a lubricant and shock absorber (it enables smooth gliding of the joint surfaces and, due to its viscoelastic properties, cushions the pressure generated under load). In addition, MONOVISC® is integrated into the cartilage matrix and stimulates the body’s own production of hyaluronic acid.


